To My Fellow Shareholders,
2018 was another outstanding year for Amgen. Through our progress in serving patients around the world with innovative medicines to treat serious diseases, we achieved record levels of financial performance and provided an attractive return to our shareholders.
Total revenues in 2018 were our highest ever, a record $23.7 billion, up 4% from 2017. We also achieved record non-GAAP earnings per share of $14.40,1 up 14% from the prior year, and free cash flow of $10.6 billion,1 another record. Amgen's non-GAAP operating margin of 53%,1 though down slightly from the prior year, still ranked among industry leaders. We invested $3.7 billion1 in research and development, reflecting both our commitment to innovation and our confidence in the potential of Amgen's growing pipeline.
With unwavering commitment, Amgen staff delivered a one-year total shareholder return of 15% and a five-year return of 93%, outperforming our peer group and the S&P 500 over both time periods. We also returned $21.4 billion to shareholders in 2018 in the form of dividends and share repurchases.
BUILDING OUR CAPACITY TO INNOVATE AND GROW
Our achievements in 2018 capped a five-year period of intense transformation during which we met or exceeded the ambitious financial commitments that we made to our shareholders (see below). Simultaneously—and critically—we continued to set the stage for our future, strengthening Amgen's long-term ability to innovate, compete and grow.
During our transformation, we generated approximately $1.9 billion in gross cost savings that we redeployed into our business while embedding a set of productivity capabilities throughout our organization. These capabilities open new options for Amgen's business as we have built foundational flexibility in what is a highly dynamic operating environment.
Let me share some additional highlights of our transformation, framed through our major 2018 achievements.
Innovation continues to be the lifeblood of our business. Over the past five years, we invested nearly $19 billion1 in R&D and, as a result, are now advancing a record number of clinical assets through our pipeline. This includes a blossoming early-stage oncology pipeline with approximately 20 molecules in development. For example, we put 10 new, first-in-human molecules in our portfolio in 2018 alone, and a number of those in our oncology portfolio will be generating important data over the course of 2019. In the last two years, we filed 20 investigational new drug applications, 70% of which are first-in-class, which means they are based on a new mechanism of action.
In the later stages of development, three programs have reached Phase 3, including tezepelumab, a first-in-class biologic treatment for severe asthma. In 2018, the U.S. FDA granted a Breakthrough Therapy Designation for tezepelumab in a significant subset of asthma patients—those without an eosinophilic phenotype. The unmet need for asthma is enormous, and new treatments are urgently needed. Severe asthma affects more than 30 million people worldwide and is expected to grow as more people move to cities.
Along with our substantial R&D investment, we made significant strides in R&D productivity. During our transformation, we drove a 3-percentage-point improvement in R&D efficiency—meaning that the output we are achieving today with our R&D investment at 16%1 of sales is roughly equivalent to what we were achieving with 19%1 of our sales five years ago. In short, we are achieving more with each dollar of investment.
Our R&D cycle times also decreased, further improving our efficiency. During the past five years, we reduced our new drug development cycle time by an average of approximately 36 months, reflecting our commitment to bringing innovative medicines to patients as quickly and safely as possible. One prominent example: in 2018, we moved AMG 510, a potential treatment for non-small cell lung and colorectal cancers, from discovery to first-in-human trials in just seven months. That was a record for us, and we expect to have new clinical data to share on AMG 510 later in 2019.
2014-2018
We also continue to invest heavily in what we believe is a differentiated approach to human genetics. We now have a pipeline that is 75% genetically validated to various degrees, up from 15% five years ago. Human genetic validation provides us with earlier, better insights into which of the proposed new therapies have the greatest potential for approval and success, enabling us to invest accordingly.
Amgen took an early lead in human genetic validation in 2012 by acquiring Iceland-based deCODE Genetics, now a wholly owned Amgen subsidiary. Today, more than half of the Icelandic population are participants in deCODE's research, along with 1 million participants outside of Iceland. In 2018, we expanded this capability through a new collaboration with a prominent U.S.-based integrated delivery network. This should bring us an additional 500,000 participants to study by genotype and phenotype.
Our R&D pipeline yielded nine approved and launched products during our transformation, including two in new therapeutic areas for Amgen—Repatha® in cardiovascular disease (launched in 2015) and Aimovig® in neuroscience (launched in collaboration with Novartis in 2018).
Repatha® is an LDL cholesterol treatment proven to reduce heart attacks and strokes in patients with established heart disease. Repatha® sales increased 72% in 2018 to $550 million, with volume nearly doubling versus the prior year. In 2018, we made Repatha® available at a 60% reduced list price in the U.S. to help lower out-of-pocket costs, especially for Medicare patients, which will enable us to serve more patients (see below).
Aimovig® emerged as one of the most successful new product introductions in recent industry history. Aimovig® is purpose-built to treat migraine and is quickly gaining acceptance among patients and physicians. With millions of migraine patients in the U.S. alone, we expect Aimovig® to be a significant growth driver for Amgen for years to come.
In 2018, we launched Parsabiv®, the first new treatment in more than a decade for secondary hyperparathyroidism. This therapy provides patients and healthcare providers with a novel option to help treat a complex disease that affects a significant number of patients on hemodialysis.
Our oncology/hematology portfolio is now well positioned to be an important growth engine for the future. In 2018, combined sales of XGEVA®, KYPROLIS®, Nplate®, Vectibix®, BLINCYTO® and IMLYGIC® exceeded $4.4 billion, growing 14% over the prior year. KYPROLIS®, one of our newer medicines, has delivered a suite of clinical evidence demonstrating its ability to meaningfully extend overall survival in patients with relapsed or refractory multiple myeloma.
Prolia®, prescribed to reduce the risk of fracture in postmenopausal women with osteoporosis, continued to deliver strong double-digit volume growth in 2018. Launched in 2010, it is being used to treat approximately 4 million people, a fraction of the tens of millions of people who have osteoporosis worldwide and are at risk from the often devastating effects of bone fracture.
2018 was also a landmark year in our commitment to biosimilars, a business we established during our transformation period. Biosimilars can offer new, high-value options for patients and payers, and Amgen is committed to success in this promising but highly competitive arena. In 2018, we launched our first biosimilars, both outside the U.S.: AMGEVITA™, a biosimilar to AbbVie's HUMIRA®, and KANJINTI™, a biosimilar to Roche's Herceptin®. We believe that Amgen's reputation for reliable supply and safety is an important contributor to our success in this field. We plan to launch a series of biosimilars in the coming years, taking advantage of our deep expertise and long, high-quality track record in precision biologics manufacturing.
Further leveraging this expertise, we developed and deployed our next-generation biomanufacturing capability. During our transformation, we advanced two state-of-the-art facilities, which improve both efficiency and environmental friendliness, while sustaining our high standards for quality and reliable supply. Our first next-generation facility, in Singapore, was licensed by regulators in 2017. Last year, we broke ground on a second, similar facility in Rhode Island, where we already have a large manufacturing presence. This investment became possible due to improved U.S. competitiveness from tax reform.
Reliable supply is an essential element in the drive to expand Amgen's global commercial reach. We now have a presence in approximately 100 countries, up from 50 countries in 2011, enabling us to play a growing role in serving the world's exploding demand for better healthcare. In January 2019, we launched Repatha® in China, our inaugural product launch in what is now the world's second largest pharmaceutical market. In Japan, the world's third largest pharma market, the Amgen Astellas BioPharma joint venture recently received two important new product approvals: BLINCYTO® for the treatment of relapsed or refractory B-cell acute lymphoblastic leukemia and EVENITY™† for the treatment of osteoporosis in patients at high risk of fracture.
We fully recognize the challenge of global expansion and are greatly encouraged by our strong unit volume growth internationally. Sales from outside the U.S. are expected to account for an increasing percentage of our total revenues over the next decade.
COMMITTED TO LONG-TERM GROWTH
Success in the research-based biopharmaceutical industry demands the ability to deliver results not just quarter to quarter but generation to generation as well. As just discussed, we have numerous meaningful opportunities ahead, including recently launched branded biologic and biosimilar products, our continued expansion into new markets globally and the cultivation of a robust and differentiated pipeline. We have also anticipated challenges in front of us, including generic competition for Sensipar®, increasing competition for our rheumatoid arthritis product Enbrel® and additional biosimilar competition for EPOGEN®, NEUPOGEN® and Neulasta®.
We are prepared for these challenges and face them from a position of strength. I want to make it clear: we have a long-term commitment to the hallmark of Amgen—innovation—and have the determination and resources to meet these competitive challenges and deliver growth over the long term.
Notably, we now have the ability to operate more quickly and nimbly across the business, starting in R&D and extending through manufacturing and commercial operations. We are a leader in capital-efficient biologics manufacturing and have built a global commercial enterprise focused on cultivating the best opportunities, wherever they may be. We have a strong balance sheet and cash flows, a reputation for excellence in science and strategic partnership and a culture that is a beacon for people with the sophisticated knowledge and skill to make a difference in global health. And we are well positioned with a broad array of products that address large patient populations in areas of great unmet need, along with therapies that make a difference for more limited patient populations.
In short, while our ambitious five-year goals for 2018 were reached, our transformation continues. We have great confidence in our ability to meet the expected challenges and to break through to the next phase of growth.
BENDING THE CURVE OF HEALTHCARE
In less than 40 years, Amgen went from a tiny, venture-backed biotechnology startup to an important force in global healthcare. We are now integrated into the global healthcare ecosystem, centered on patients but also engaging fully with other major healthcare communities: physicians and other providers, governments and private payers, investors, regulators and society at large.
In 2018, we began talking in earnest about our long-term ambition: to bend the curve of healthcare. By that, we're signaling that we intend to take a significant role to ensure that more people have greater access to better healthcare, both now and for generations to come. We are committed to exploring and pursuing healthcare solutions that reinvent and even disrupt the current approaches to healthcare access, cost, value and innovation.
The galvanizing call to "bend the curve of healthcare" is rooted in the recognition that healthcare systems globally are at an inflection point. People want better healthcare and are striving to secure it. While we continue to see exciting opportunities ahead, the Age Wave is cresting with unprecedented challenges to keeping people healthy and productive.
Many healthcare systems are feeling the strains, though they are responding to today's problems with yesterday's systems and solutions. In the U.S., people know intuitively that the healthcare system is cumbersome and the upward curve of cost is not sustainable.
We are sensitive to this sentiment. Overall, the net selling price of our medicines declined in 2018, and we expect a mid-single-digit decline in 2019. In general, we believe sales growth will ultimately come from increased volume and access as opposed to price increases. We are also committed to helping patients access our medicines via the Amgen Safety Net Foundation, which provides our therapies to qualifying U.S. patients who cannot afford them. In 2018, the commercial value of our therapies provided through the Amgen Safety Net Foundation exceeded $1 billion. We also made a donation of Amgen cancer treatments and supportive care medicines, commercially valued at $93 million, to nonprofit, humanitarian aid organization Direct Relief, which they distributed to patients in 18 developing countries at zero cost.
Drug costs, however, are just part of the picture, given that biopharmaceuticals represent approximately 14% of the U.S. healthcare bill, roughly the same percentage as in 1960. Study after study has shown that good health is strongly connected with human happiness and productivity and that it is also a galvanizing force for societal and economic development. At Amgen, bending the curve in healthcare means finding ways, including disruptive ones, to accelerate the positive trends in healthcare and change the course of negative ones.
Consider cardiovascular disease as an example. The good news here is that due to advances in prevention, public health and medicine, the rates of cardiovascular disease have dropped precipitously over the past 20 years. The bad news is that despite decades of steady progress, half of all adults in the U.S. still have some type of cardiovascular disease. In 2016 (the last year for which we have statistics), the number of deaths from cardiovascular disease actually rose over the prior year to approximately 840,000 in the U.S. Behind that statistic are millions of heartbreaking stories—from people debilitated by failing hearts to family caregivers who must now face difficult choices. Aside from the human misery, there's a staggering direct cost: some $600 billion in the U.S. alone, as projected by the American Heart Association for 2019. This is slated to rise to an estimated $900 billion by 2030 if we, as a society, fail to intercept this trajectory.
We can bend the curve. It will take much in the way of partnership, including coordinated efforts in patient education, healthcare access, prevention and public health. It will also take continued biomedical innovation, such as the science behind Repatha®, to meet the needs of those for whom current treatments in lipid lowering may not be enough.
At a time when someone in the U.S. has a heart attack or stroke every 40 seconds, a medicine such as Repatha® can bend the curve of healthcare in the right direction. That is why we work so hard to ensure that every patient who might benefit from Repatha® has access to it. It is also why we are working on a new treatment for heart failure, omecamtiv mecarbil, in Phase 3 clinical development.
Overall, a rapidly aging global population will mean a dramatic rise in the number of people suffering from serious, age-related illnesses like cancer, dementia and cardiovascular disease. Medicare and Medicaid direct costs for Alzheimer's are estimated at $186 billion annually—rising to $750 billion by 2050 if the current trends continue. Our effort in this area includes AMG 520, a Phase 3 asset we are developing in partnership with Novartis, which could provide a new preventative treatment option.
Innovation has genuine potential to bend the curve of Alzheimer's disease and many other serious diseases that impact millions of people. We know we must continue to innovate—and frame the power of innovation in our dialogue with others—so that patients today and in the future benefit from the biomedical revolution we are helping to shape.
That dialogue starts with listening. We listen intently to all healthcare communities, starting with patients, who visit our facilities to speak with, teach and inspire us (see below). There is nothing more powerful for us than to listen to patients whose lives have been meaningfully affected by our work—and whose own life trajectories have been changed through our efforts and your investment.
We know that for every patient success story, there are still far too many others that don't end well. "Bending the curve of healthcare" takes on special meaning when many patients do not have time on their side. That realization brings a sense of urgency to everything we do.
WE ARE EXCITED ABOUT THE FUTURE
Looking ahead, we are ready to build on our recent transformation successes, including maintaining and improving on our leading process development and manufacturing capabilities, driving our innovation and expanding our outreach in patient access and services to more regions around the world. We will sustain the gains of the past five years through a culture of productivity so that the time, talent and capital invested in Amgen is put to good use. We will continue to deliver on our commitments to patients, to you as shareholders and to all those we serve, both year to year and generation to generation.
On behalf of Amgen's board of directors and leadership team, thank you for your continued investment and support. I also thank our more than 21,000 staff members around the world for their commitment to ethics—and to our mission. It is our shared privilege to work at the leading edge of innovation and for a company making a positive difference in the lives of so many people every day.
Robert A. Bradway
Chairman and Chief Executive Officer
EVERY 40 SECONDS...

Every 40 seconds, someone in the U.S. has a heart attack or stroke. Repatha®, Amgen's medicine for people with high LDL cholesterol who are at risk for heart attacks and strokes, can help to address this significant public health issue, but concerns over out-of-pocket costs were proving to be a barrier to its use for a large number of patients. For example, an estimated 75% of Medicare patients prescribed a PCSK9 inhibitor like Repatha® were never actually filling their prescriptions—a situation that we found unacceptable.
In October 2018, Amgen took the bold action of making Repatha® available in the U.S. at 60% less than its original list price, leading directly to lower out-of-pocket costs for some patients, especially seniors on Medicare. Bob C. from Boca Raton, Florida, shown here, is one of them.
"Since turning 65 and going on Medicare, I have had to make one of the hardest decisions of my life: can I afford to stay on Repatha®, the only thing—even after nine stents and numerous statins—that has worked to lower my cholesterol?" he said. "Amgen's decision to significantly lower the list price of Repatha® means that I can continue taking a therapy that has been so effective for me."
Amgen's action drew praise from the American Heart Association, which stated, "We are heartened by Amgen's substantial commitment to reduce the costs of their proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor as part of a larger collaborative effort across the public, private and independent sectors to shift the focus to the value of treatment and not simply its costs."
BRINGING BITE TO THE FIGHT™
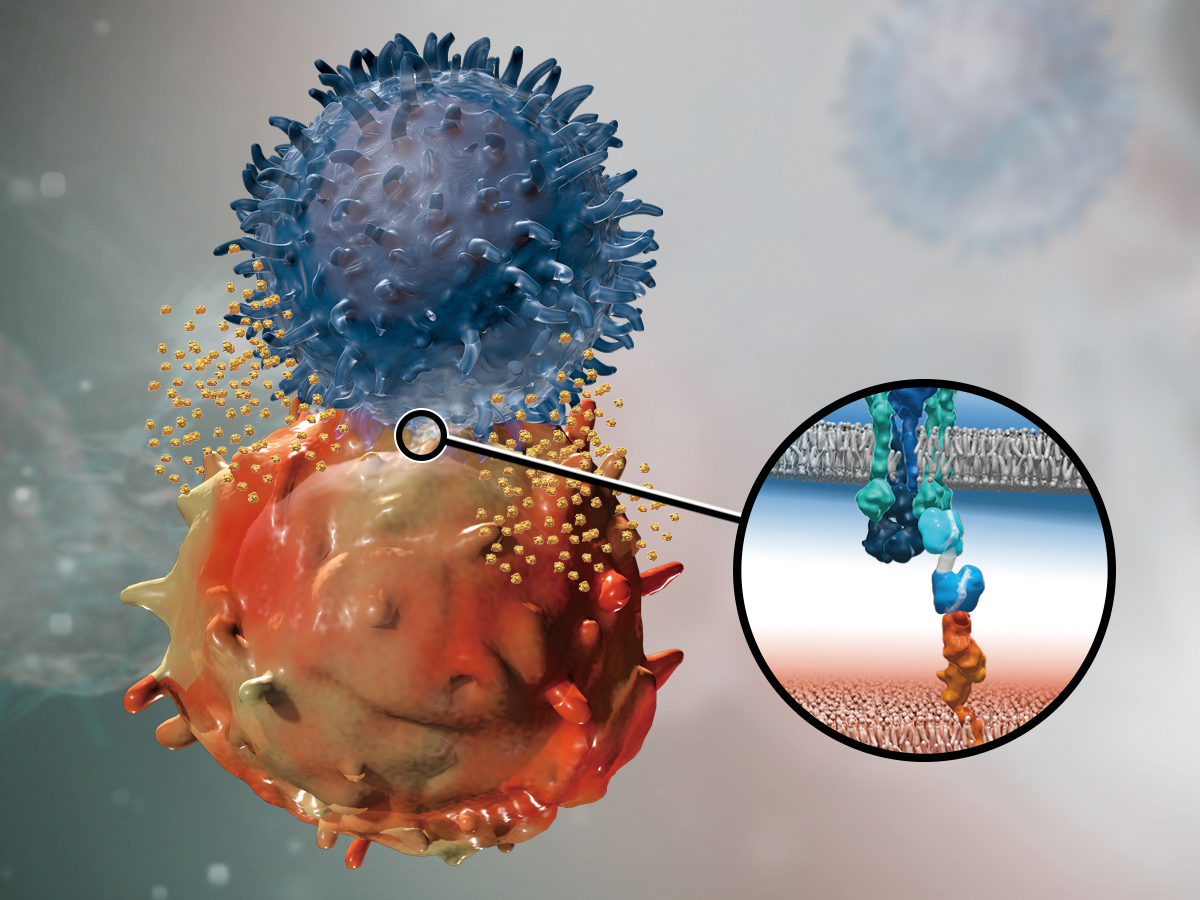
One of the great medical success stories of the past few decades has been the progress that has been made against cancer. From its peak in 1991, the cancer death rate in the U.S. has fallen by 26%, while the chances that a cancer patient will live five or more years have increased by 41% across all cancers. A large part of these remarkable gains can be attributed to innovative new cancer medicines, but we certainly can't declare victory yet over a disease that still claims more than 9 million lives a year worldwide.
In 2018, much of the attention of the oncology community was focused on a new class of medicines known as bispecific T cell engagers, or BiTE® molecules. Pioneered by Amgen, BiTE® technology is a targeted immuno-oncology platform designed to engage the patient's own immune system to fight cancer.
Amgen already has one BiTE® therapy on the market: BLINCYTO® for acute refractory or relapsed B-cell lymphoblastic leukemia.
At the American Society of Hematology's annual meeting in December 2018, we presented data on two investigational BiTE® therapies in our early-stage pipeline: AMG 420 to treat patients with relapsed and/or refractory multiple myeloma and AMG 330 to treat patients with relapsed or refractory acute myeloid leukemia.
The U.S. Food and Drug Administration (FDA) has granted Fast Track and Orphan Drug Designation to AMG 420, indicating the acute need for new treatments for multiple myeloma patients, many of whom have run out of available options. "Despite recent treatment advances, multiple myeloma continues to be a disease characterized by cycles of relapse and recurrence requiring additional therapies to help control the disease," said Max S. Topp, M.D., professor, Hospital of Wuerzburg, Germany, and AMG 420 clinical study investigator.
GETTING TO A GOOD PLACE

Migraine patient Kayleen H. (right) met Amgen scientist Cen Xu (left), who led the team that discovered and developed Aimovig®.
At the start of every year, Amgen gathers its top leaders from around the world to gain alignment on our path forward. The highlight of these meetings is the opportunity to hear firsthand from patients who have benefited from our medicines, and this year's meeting was no exception.
Kayleen H. has been battling migraine for more than 20 years. Like so many others with this misunderstood and undertreated disease, Kayleen suffered frequent and debilitating migraines that came to dominate her life, preventing her from working and placing a tremendous burden on her family. In fact, according to Kayleen, her migraine on a scale of one to 10 was an eight—each and every day—and often spiked higher than that.
As was the case with Kayleen, migraine frequently strikes people in the prime of their lives. As a result, migraine is particularly costly in terms of lost productivity, with U.S. employers losing an estimated $11 billion each year, largely due to missed workdays.
Kayleen tried many treatment options over the years with little success. She began taking Aimovig®, Amgen's new medicine specifically designed to help prevent migraine, in June 2018. "I have been amazed at how well it works," she said. "I have less migraine days now, and I'm in a really good place."‡
EXTENDING A HELPING HAND IN OUR OWN BACKYARD

(From left to right) John Saffelo, Global Security Manager, Amgen; Lt. Jennifer Seetoo, LASD Malibu/Lost Hills Station; Nick Razum, Global Security Director, Amgen; Will Dobbins, Global Security Manager, Amgen; Dave Barley, Global Security Director, Amgen and Dep. Bart Marshall, LASD Temple Station
Over the years, Amgen has helped communities around the world to recover after natural disasters. In 2018, disaster struck close to home. On November 7, a gunman killed 12 people, including many college students, at the Borderline Bar and Grill, located less than four miles from our world headquarters in Thousand Oaks, California. The next afternoon, as our community was reeling from this terrible act of violence, major wildfires broke out and raged in the valleys surrounding our campus for two weeks before they were finally contained. While thankfully no Amgen lives were lost, thousands of our staff were forced to gather their belongings and flee their homes.
Our Amgen campus was closed for several days to all but essential personnel, including about 80 staff assigned to our Emergency Operations Center. These dedicated colleagues rotated in 12-hour shifts for nine days throughout the crisis. They assessed threats to our buildings, monitored air quality to determine the health risk posed by the pervasive smoke and kept track of the safety of our people. Beyond this core team, hundreds of Amgen staff lent a helping hand by giving blood, donating to relief funds and opening their own homes to colleagues and neighbors who were evacuated.
We showed our appreciation to local first responders by sending multiple food trucks to the Woolsey Fire Command Post and other field operations, where professionals and volunteers were serving shifts as long as 18 hours to contain the fires. Amgen staff expressed their gratitude to these heroes with hundreds of thank you notes as well as events for the first responders and their families.
In addition to these personal expressions of support, the Amgen Foundation pledged $500,000 to support victims of the fires and $250,000 to support those affected by the Borderline shooting.
BUILDING A WINNING CULTURE
Of the thousands of companies founded as part of the biotech revolution of the 1980s, only two of note remain independent to this day. One of them is Amgen.
We believe the key to our enduring success is a culture built around a mission to serve patients, an aspiration to be the world's best human therapeutics company and a set of values that emphasize patients, science, ethics, trust and respect.
Our mission, aspiration and values resonate with our staff. In fact, we regularly survey our staff and are encouraged that 95% understand how their work contributes to Amgen's success. Our surveys also ask what three words come to mind when thinking about Amgen's culture. The words cited most often are "innovative," "patients" and "science." We do not take our culture for granted. With unemployment in the U.S. at or near all-time lows and more than 20,000 job openings across peer companies in our industry, we understand that talented people—"whether they have decades of experience or are just starting their careers—"have more options than ever before.
To ensure that Amgen staff continue to choose Amgen as an employer, we have introduced several staff and leadership development programs in recent years and enhanced our benefits to support employees in their life experiences. These include programs that coach families through college decisions, ship milk home for traveling moms and help with care for an aging parent.
Ensuring that all staff feel a sense of belonging in the company is essential to our success. To this end, thousands of our staff members participate in our diversity-focused employee resource groups that help them navigate their careers, while also providing opportunities to contribute to our business and the community. Each of these groups is sponsored by a leader who reports directly to the CEO.
And these efforts to build a winning culture are not just felt by our teams but are being recognized externally as well.
In 2018, Amgen earned a spot on several "best" lists compiled by Forbes magazine: "America's Best Large Employers," "America's Best Employers for Diversity" and "America's Most Just Companies." The career website Indeed named Amgen one of its "Best Places to Work in Healthcare," with another site, CareerBliss, ranking us as one of the "50 Happiest Companies in America." The magazine G.I. Jobs named Amgen as a top "Military Friendly Employer," while, for the second year in a row, we received a perfect score on the Human Rights Campaign Foundation's "Corporate Equality Index," which rates companies on their policies and practices pertinent to lesbian, gay, bisexual, transgender and queer (LGBTQ) staff.
Amgen also received multiple international recognitions throughout the year for its culture and sustainable practices. Our Europe hub was certified by Great Place to Work. Amgen Italy was recently certified as one of the seven Best Companies for Gender Equality by Winning Women Institute. Amgen Australia was awarded the Australian Packaging Covenant Award (Pharmaceutical Category), which is focused on more sustainable packaging throughout the supply chain to reduce the environmental impact of packaging in Australia. Amgen China received the "Top 10 Pharmaceutical Pioneers" award at the Annual Medical Experts Summit. And Amgen Singapore Manufacturing received the Grand Winner award for Best Bioprocessing Excellence in Asia.

In June 2018, staff from our LGBTQ employee resource group celebrated PRIDE month with a flag-raising event at our site in Cambridge, Massachusetts.
- This is a non-GAAP financial measure. See reconciliation to U.S. generally accepted accounting principles (GAAP) accompanying this letter.
- On average, between 2013-2018
- On average, between 2014-2018
- EVENITY™ is developed in collaboration with UCB globally, as well as our joint venture partner Astellas in Japan. EVENITY™ trade name provisionally approved by the FDA.
- Kayleen's story is unique; the experiences of other Aimovig® patients may vary.
Reconciliations of GAAP to Non-GAAP Measures (Unaudited)
(In millions, except per share data)
| Years ended December 31, | 2018 | 2017 | 2016 | 2015 | 2014 | 2013 |
|---|---|---|---|---|---|---|
| GAAP research and development expenses | $3,737 | $3,562 | $3,840 | $4,070 | $4,297 | $4,083 |
| Adjustments to research and development expenses: | ||||||
| Acquisition-related expenses(a) | (78) | (77) | (78) | (89) | (124) | (142) |
| Certain net charges pursuant to our restructuring initiative(b) | (2) | (3) | (7) | (64) | (49) | — |
| Other | — | — | — | — | (3) | (12) |
| Total adjustments to research and development expenses | (80) | (80) | (85) | (153) | (176) | (154) |
| Non-GAAP research and development expenses | $3,657 | $3,482 | $3,755 | $3,917 | $4,121 | $3,929 |
| GAAP operating income | $10,263 | $9,973 | $9,794 | $8,470 | $6,191 | $5,867 |
| Adjustments to operating income: | ||||||
| Acquisition-related expenses(a) | 1,557 | 1,594 | 1,510 | 1,377 | 1,546 | 986 |
| Certain net charges pursuant to our restructuring and other cost savings initiatives(b) | 12 | 88 | 37 | 114 | 596 | 71 |
| Expense (benefit) related to various legal proceedings | — | — | 105 | 91 | (3) | 14 |
| Expense resulting from clarified guidance on branded prescription drug fee(c) | — | — | — | — | 129 | — |
| Other | 25 | 3 | — | — | 16 | 34 |
| Total adjustments to operating income | 1,594 | 1,685 | 1,652 | 1,582 | 2,284 | 1,105 |
| Non-GAAP operating income | $11,857 | $11,658 | $11,446 | $10,052 | $8,475 | $6,972 |
| GAAP operating income as a percentage of product sales (operating margin) | 45.5% | 45.8% | 44.7% | 40.4% | 32.0% | 32.3% |
| Adjustments to operating income | 7.1 | 7.7 | 7.6 | 7.6 | 11.9 | 6.0 |
| Non-GAAP operating income as a percentage of product sales (operating margin) | 52.6% | 53.5% | 52.3% | 48.0% | 43.9% | 38.3% |
| GAAP net income | $8,394 | $1,979 | $7,722 | $6,939 | $5,158 | $5,081 |
| Adjustments to net income: | ||||||
| Adjustments to operating expenses | 1,594 | 1,685 | 1,652 | 1,582 | 2,284 | 1,105 |
| Adjustments to other income(d) | (68) | — | — | — | — | 34 |
| Income tax effect of the above adjustments(e) | (362) | (538) | (525) | (496) | (717) | (376) |
| Other income tax adjustments(f) | 15 | 6,120 | (64) | (71) | (25) | (30) |
| Total adjustments to net income: | 1,179 | 7,267 | 1,063 | 1,015 | 1,542 | 733 |
| Non-GAAP net income | $9,573 | $9,246 | $8,785 | $7,954 | $6,700 | $5,814 |
| Weighted-average shares for GAAP diluted EPS | 665 | 735 | 754 | 766 | 770 | 765 |
| Weighted-average shares for Non-GAAP diluted EPS* | 665 | 735 | 754 | 766 | 770 | 765 |
| GAAP diluted EPS | $12.62 | $2.69 | $10.24 | $9.06 | $6.70 | $6.64 |
| Non-GAAP diluted EPS | $14.40 | $12.58 | $11.65 | $10.38 | $8.70 | $7.60 |
- Dilutive securities used to compute Non-GAAP diluted EPS for the year ended December 31, 2013 were computed under the treasury stock method assuming that we do not expense stock options.
- The adjustments related primarily to noncash amortization of intangible assets acquired in business combinations. For the years ended December 31, 2018 and 2017, the adjustments to operating income also included impairments of intangible assets acquired in business combinations.
- The adjustments related to headcount charges, such as severance, and to asset charges, such as asset impairments, accelerated depreciation and other charges related to the closure of our facilities.
- The adjustment related to the recognition of an additional year of the nontax deductible branded prescription drug fee, as required by final regulations issued by the Internal Revenue Service.
- For the year ended December 31, 2018, the adjustment related to the net gain associated with the Kirin-Amgen share acquisition.
- The tax effect of the adjustments between our GAAP and non-GAAP results takes into account the tax treatment and related tax rate(s) that apply to each adjustment in the applicable tax jurisdiction(s). Generally, this results in a tax impact at the U.S. marginal tax rate for certain adjustments, including the majority of amortization of intangible assets, whereas the tax impact of other adjustments, including restructuring expense, depends on whether the amounts are deductible in the respective tax jurisdictions and the applicable tax rate(s) in those jurisdictions.
- The adjustments related to certain acquisition items and prior-period items excluded from GAAP earnings. For the year ended December 31, 2017, the adjustment related primarily to the impact of U.S. Corporate tax reform, including the repatriation tax on accumulated foreign earnings and the remeasurement of certain net deferred and other tax liabilities.
Reconciliations of Cash Flows (Unaudited)
(In millions)
| Year ended December 31, | 2018 |
|---|---|
| Net cash provided by operating activities | $11,296 |
| Net cash used in investing activities | 14,339 |
| Net cash used in financing activities | (22,490) |
| Increase in cash and cash equivalents | 3,145 |
| Cash and cash equivalents at beginning of year | 3,800 |
| Cash and cash equivalents at end of year | $6,945 |
| Net cash provided by operating activities | $11,296 |
| Capital expenditures | (738) |
| Free cash flow | $10,558 |
Reconciliation of GAAP to Non-GAAP Financial Measures
The non-GAAP financial measures are derived by excluding certain amounts, expenses or income, from the corresponding financial measures determined in accordance with GAAP. The determination of the amounts that are excluded from these non-GAAP financial measures is a matter of management judgment and depend upon, among other factors, the nature of the underlying expense or income amounts recognized in a given period. Historically, management has excluded the following items from these non-GAAP financial measures, and such items may also be excluded in future periods and could be significant:
- Expenses related to the acquisition of businesses, including amortization and/or impairment of acquired intangible assets, including in-process research and development, adjustments to contingent consideration, integration costs, severance and retention costs and transaction costs;
- Charges associated with restructuring or cost saving initiatives, including but not limited to asset impairments, accelerated depreciation, severance costs and lease abandonment charges;
- Legal settlements or awards;
- The tax effect of the above items;
- Non-routine settlements with tax authorities; and
- The impact of the adoption of the U.S. Corporate tax reform.
Forward-Looking Statements: This communication contains forward-looking statements that are based on the current expectations and beliefs of Amgen. All statements, other than statements of historical fact, are statements that could be deemed forward-looking statements, including estimates of revenues, operating margins, capital expenditures, cash, other financial metrics, expected legal, arbitration, political, regulatory or clinical results or practices, customer and prescriber patterns or practices, reimbursement activities and outcomes and other such estimates and results. Forward-looking statements involve significant risks and uncertainties, including those discussed below and more fully described in the Securities and Exchange Commission reports filed by Amgen, including our most recent annual report on Form 10-K and any subsequent periodic reports on Form 10-Q and current reports on Form 8-K. Unless otherwise noted, Amgen is providing this information as of the date of this news release and does not undertake any obligation to update any forward-looking statements contained in this document as a result of new information, future events or otherwise.
No forward-looking statement can be guaranteed and actual results may differ materially from those we project. Our results may be affected by our ability to successfully market both new and existing products domestically and internationally, clinical and regulatory developments involving current and future products, sales growth of recently launched products, competition from other products including biosimilars, difficulties or delays in manufacturing our products and global economic conditions. In addition, sales of our products are affected by pricing pressure, political and public scrutiny and reimbursement policies imposed by third-party payers, including governments, private insurance plans and managed care providers and may be affected by regulatory, clinical and guideline developments and domestic and international trends toward managed care and healthcare cost containment. Furthermore, our research, testing, pricing, marketing and other operations are subject to extensive regulation by domestic and foreign government regulatory authorities. We or others could identify safety, side effects or manufacturing problems with our products, including our devices, after they are on the market. Our business may be impacted by government investigations, litigation and product liability claims. In addition, our business may be impacted by the adoption of new tax legislation or exposure to additional tax liabilities. While we routinely obtain patents for our products and technology, the protection offered by our patents and patent applications may be challenged, invalidated or circumvented by our competitors, or we may fail to prevail in present and future intellectual property litigation. We perform a substantial amount of our commercial manufacturing activities at a few key facilities, including in Puerto Rico, and also depend on third parties for a portion of our manufacturing activities, and limits on supply may constrain sales of certain of our current products and product candidate development. We rely on collaborations with third parties for the development of some of our product candidates and for the commercialization and sales of some of our commercial products. In addition, we compete with other companies with respect to many of our marketed products as well as for the discovery and development of new products. Discovery or identification of new product candidates or development of new indications for existing products cannot be guaranteed and movement from concept to product is uncertain; consequently, there can be no guarantee that any particular product candidate or development of a new indication for an existing product will be successful and become a commercial product. Further, some raw materials, medical devices and component parts for our products are supplied by sole third-party suppliers. Certain of our distributors, customers and payers have substantial purchasing leverage in their dealings with us. The discovery of significant problems with a product similar to one of our products that implicate an entire class of products could have a material adverse effect on sales of the affected products and on our business and results of operations. Our efforts to acquire other companies or products and to integrate the operations of companies we have acquired may not be successful. A breakdown, cyberattack or information security breach could compromise the confidentiality, integrity and availability of our systems and our data. Our stock price is volatile and may be affected by a number of events. Our business performance could affect or limit the ability of our Board of Directors to declare a dividend or our ability to pay a dividend or repurchase our common stock. We may not be able to access the capital and credit markets on terms that are favorable to us, or at all.



