While multispecific medicines account for just a sliver of drugs now approved, most of Amgen’s early-stage pipeline consists of molecules directed at two or more targets. “The trend toward multispecific drugs is a wave that is sweeping across the entire industry,” said Ray Deshaies, senior vice president for Global Research. “We want to make sure that Amgen remains on the leading edge of that wave.”
Amgen’s expertise in this field has been strengthened by several key acquisitions. The BiTE® (bispecific T cell engager) platform is an outgrowth of the 2012 acquisition of Micromet, now Amgen Research Munich. The 2019 acquisition of Nuevolution, now Amgen Research Copenhagen, added DNA-encoded library technology to accelerate our search for multispecific small molecules.
With the recently completed acquisition of Teneobio “we now have the most sophisticated tools available to generate the building blocks for large molecule multispecifics,” said Deshaies. “This technology isn’t only important for our T cell engagers but will benefit all of our therapeutic areas.”
“At Teneobio, we were committed to developing antibody therapies to improve the lives of patients and cure diseases,” said Katherine “Kat” Harris, who came to Amgen with the acquisition and is now vice president, Discovery. “As a small company, we already had more programs in our pipeline than we could take forward. With Amgen, we'll be able to deliver therapies at a pace and scale we couldn’t achieve on our own.”
From two chains to one
The Y-shaped antibodies produced by our immune systems have binding regions that match a specific shape. These shapes, called antigens, are usually segments of foreign or mutated proteins that are perceived as “non-self,” hence a threat. Drug designers can use the precision targeting provided by antibodies to engage many proteins involved in disease. However, it’s more difficult to make multispecific antibodies—one molecule that binds two or more antigens.
The challenge stems from the structure of antibodies, which are made up of light and heavy protein chains produced by two different genes. “When cells are assembling multispecific antibodies, the light and heavy chains don’t necessarily pair the way we want them to pair,” said Deshaies. “The cells tend to make a complete mélange of molecules with unpredictable properties.”
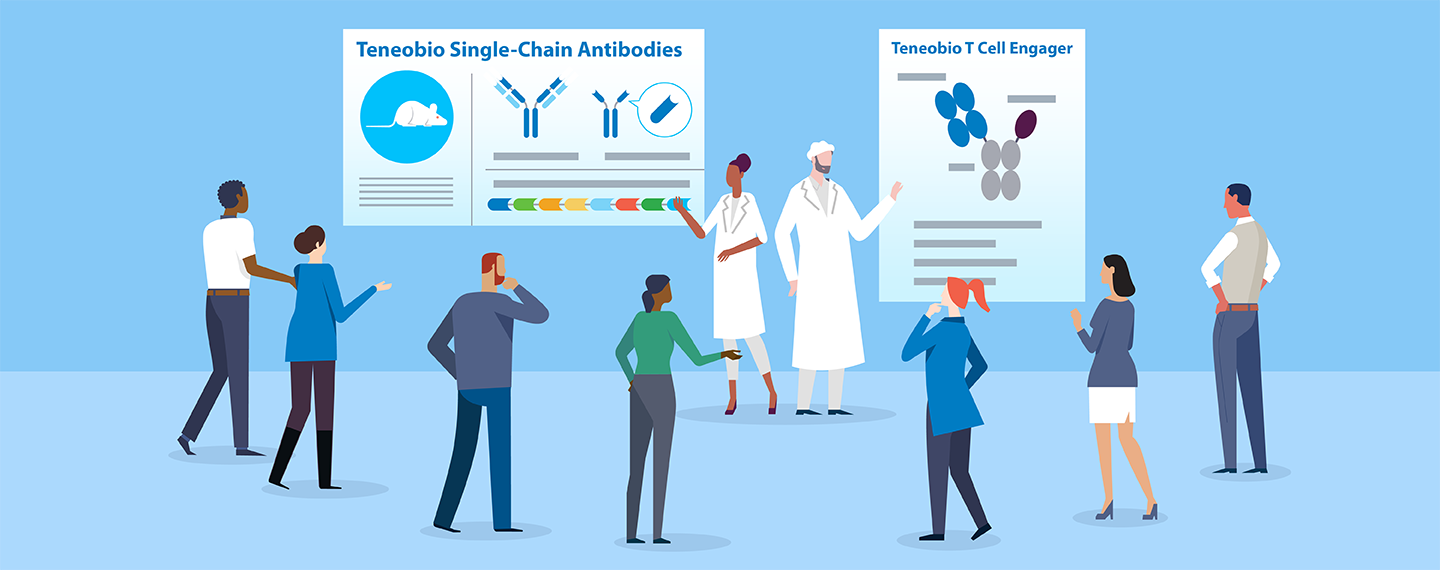
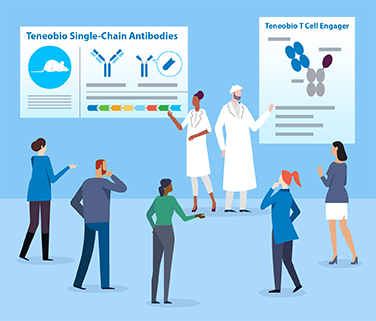
Protein engineers have devised clever ways to solve the mispairing problem, but implementing these solutions takes time, and the workarounds may not fix other issues. Teneobio has solved the mispairing problem through its UniRat® technology. The platform is based on a genetically modified rat that produces fully human UniAb® antibodies composed of a single heavy chain.
“A UniAb® molecule acts just like an antibody—it is, in fact, an antibody—so it has the same extended half-life, and good manufacturability that comes with conventional antibodies,” said Harris. “These heavy chain-only antibodies offer unparalleled flexibility in terms of developing multispecific therapies. They also allow us to use sequence-based discovery to screen very large numbers of antibody genes very quickly to identify the best therapeutic candidates.”
Turning the tortoise into a hare
It’s difficult to use sequence-based discovery with conventional antibodies because the light and heavy chains have different genes. “If we just sequence all the antibody genes from an immunized animal, it’s hard to know which light chains go with which heavy chains,” said Alan Russell, vice president, Biologics. “By getting rid of the light chain, we can think about discovery in a digitally transformed way. We can turn the tortoise into a hare.”
Scientists at Teneobio have used sequencing to sort through large repertoires of antibody genes, group antibodies into related sequence families and rank those most likely to have high affinity for a target antigen. “It’s a simple equation,” said Harris. “The more abundant an antibody sequence is within a sequence family, the more likely it is that the antibody will be specific for its antigen.”
Sequencing can also pinpoint some potential limitations, such as amino acid sequences associated with less stable or hard-to-manufacture antibodies. By rapidly finding good binders and weeding out problems, sequence-based screening allows scientists to focus their lab-based testing on a smaller selection of high-quality molecules. It also accelerates the job of assembling binders, like beads on a string, into multispecific drug formats.
In the long run, this new technology opens the door to dramatic improvements by facilitating in silico drug design. “Eventually, we can use computers to help us make digital libraries of antibodies and then use those libraries to understand which sequences provide the best leads,” said Russell.
To make progress, in silico methods need to work hand-in-glove with lab work in an iterative cycle. Computers can predict the best antibodies based on their sequences; scientists can make and test those antibodies, and the results can inform machine learning models and sharpen the accuracy of future predictions.
“The heavy chain-only platform will allow us to do in silico library design, which has the potential to reduce by as much as two-thirds the time it will take to develop leads and hand them off to the teams that see if those leads actually help patients,” said Russell. “That’s why we are so excited about the platform.”
New options for T cell engagers
Another Teneobio innovation could expand the therapeutic potential of Amgen’s bispecific T cell engagers. T cell engagers are designed to direct the power of the immune system against cancer by directly linking T cells to tumor cells. In some patients, the resulting immune response can lead to a cascade of events, called cytokine release syndrome (CRS), that impact normal tissue as well. While CRS can be mitigated by anti-inflammatory drugs and modified dosing regimens, these measures may reduce or delay a drug’s treatment benefits.
Teneobio has engineered a T cell engager that binds with a very low affinity to the CD3 receptor on T cells. Both high- and low-affinity T cell binders are needed in cancer therapy. Some cancer patients benefit from higher potency, even if it comes with higher cytokine release, while other patients require lower risk of CRS, even if it comes with lower potency. The addition of Teneobio will help Amgen optimize our molecules and provide the right CD3 engager for the right target, the right tumor, and the right patient.
In his comments on the day that Teneobio staff joined Amgen, Deshaies emphasized that the new combined team will be pursuing a highly ambitious research agenda.
“We are not just trying to make a few medicines, which is obviously important in and of itself. We are trying to come up with a whole new class of medicines that will completely reshape how we think about treating patients. That’s incredibly exciting.”